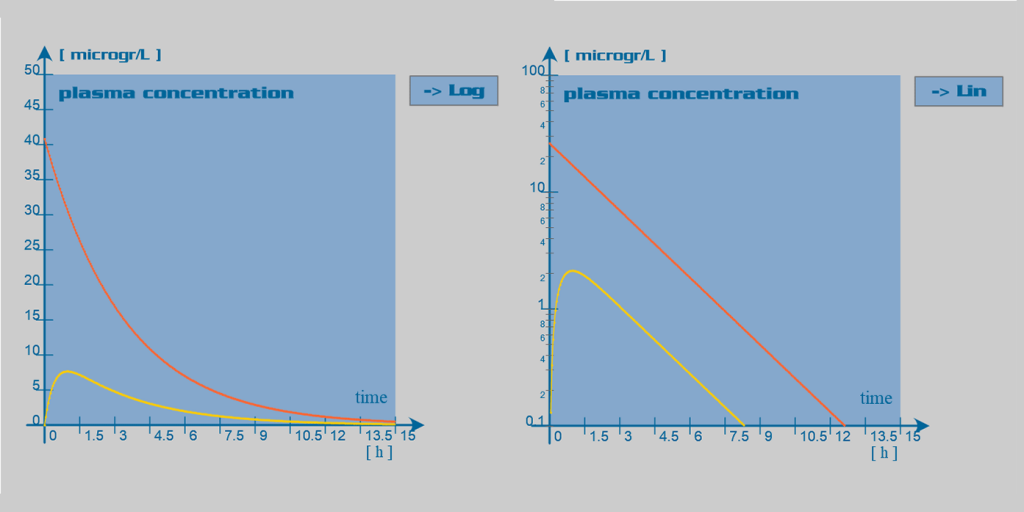
Morphine pharmacokinetic parameters
| Oral bioavailability (F) | 25% |
| Clearance (CL) | 72 L/h |
| Volume of Distribution (Vd) | 245 L |
| Half-life (t1/2) | 2.5 h |
Description
Morphine is a drug commonly used in the management of moderate to severe nociceptive pain, such as pain due to cancer, surgery or trauma.
Morphine is well absorbed through the gastrointestinal mucosa. However, it undergoes substantial hepatic first-pass effect. Therefore, its oral bioavailability is relatively low (~25%). Peak plasma drug concentrations are reached 30 to 90 min after oral administration and 15 to 20 minutes after subcutaneous or intramuscular administration. Slow release oral formulations have been developed, which deliver the dose over 8 to 12 hours (MST Continus®) or up to 24 hours (Kapanol®)
After absorption, morphine is rapidly distributed within the body, including the brain. Morphine has a large volume of distribution, and only one third of the circulating amount binds to plasma proteins.
Morphine has a high systemic clearance and an accordingly short half-life of approximately two hours. The primary site of morphine metabolism is the liver, where it undergoes rapid glucuronidation. However, extrahepatic metabolism of the drug can account for up to 30% of its total clearance. This extrahepatic metabolism may play a relatively important role in morphine metabolism in patients with severe liver failure. In the liver, the principal metabolite is the morphine-3-glucuronide (M3G). This metabolite has no pharmacological effect and is primarily excreted in the urine. Another important metabolite is morphine-6-glucuronide (M6G), which appears to be at least as potent as morphine. M6G has a half-life of approximately 1-2 hours. This active metabolite is also excreted in the urine and may accumulate significantly in case of renal insufficiency. Less than 10% of the dose is excreted unchanged in the urine.
Clinical implications
When deciding for the appropriate schedule of administration, several factors must be evaluated. The approximate relative potency of parenteral to oral morphine is 1:3. Patients with acute pain will be more readily relieved with parenteral administration of morphine (e.g iv injection, i.m or sc injection, or constant rate infusion). On the other hand considering the greater convenience of oral administration, this route is the first choice for treatment of patients with sustained pain. Changing from parenteral to oral route necessitates the administration of double to triple doses. To further increase the convenience of oral administration, controlled-release preparations have been developed, which provide prolonged analgesia for 8 to 24 hours.
Due to its short half-life, dosing must be repeated on a frequent basis (q 4 hours in general). The frequency must however be decreased in hepatic failure patients and also in renal failure patients, due to the accumulation of an active metabolite.